Smart Obstetric Forceps
Re-imagining forceps with traction feedback
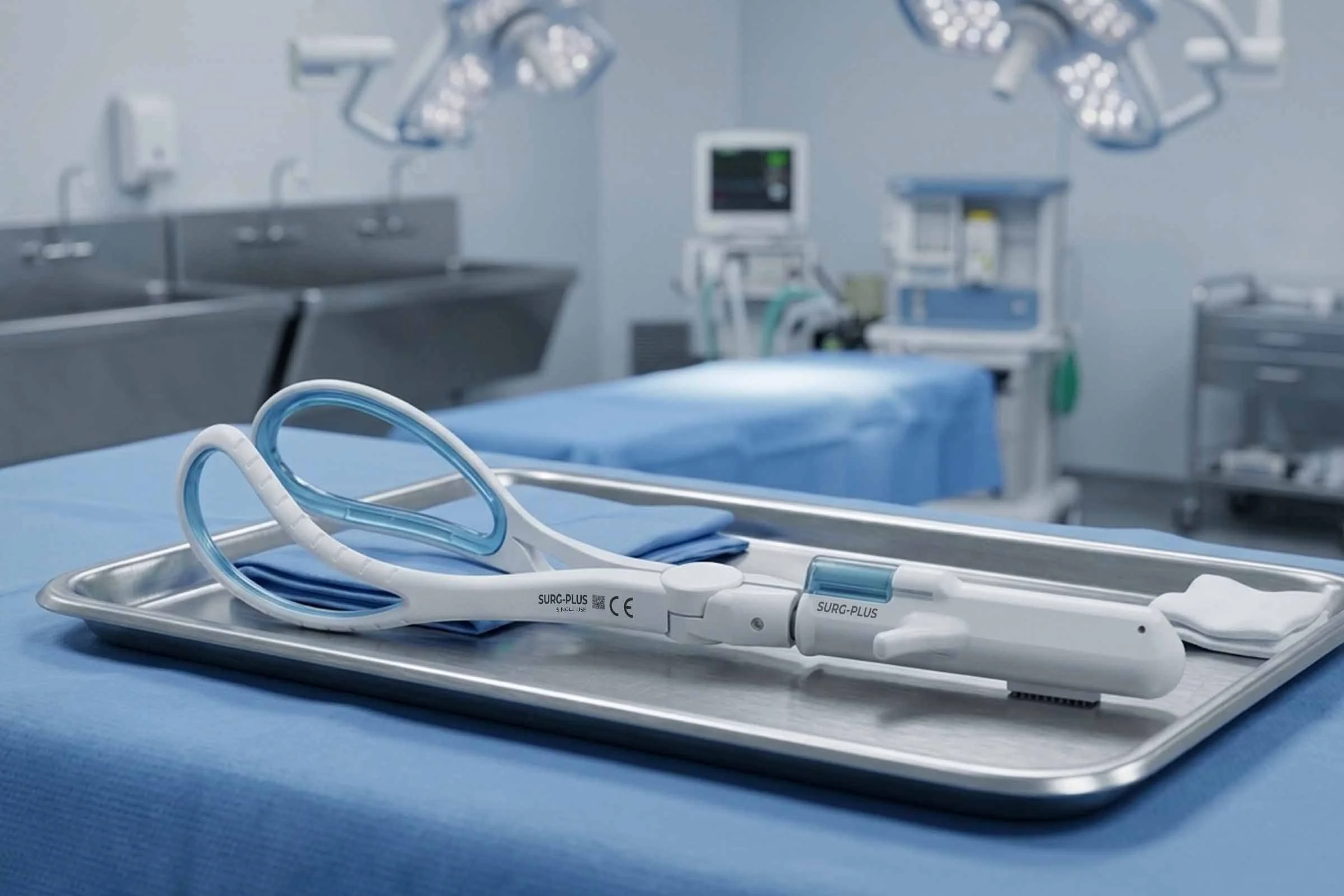
Obstetric Foreceps
Client: We collaborated with a leading obstetrician alongside Medipex and Hull and East Yorkshire NHS Trust to rethink a long-established assisted-delivery instrument. Conventional obstetric forceps have changed little in centuries and provide no feedback on traction force during use. The project aimed to improve safety and usability by introducing measurable force guidance and enhanced handling characteristics, supporting clinicians in high-pressure delivery scenarios where excessive traction can lead to avoidable trauma for mother and baby.
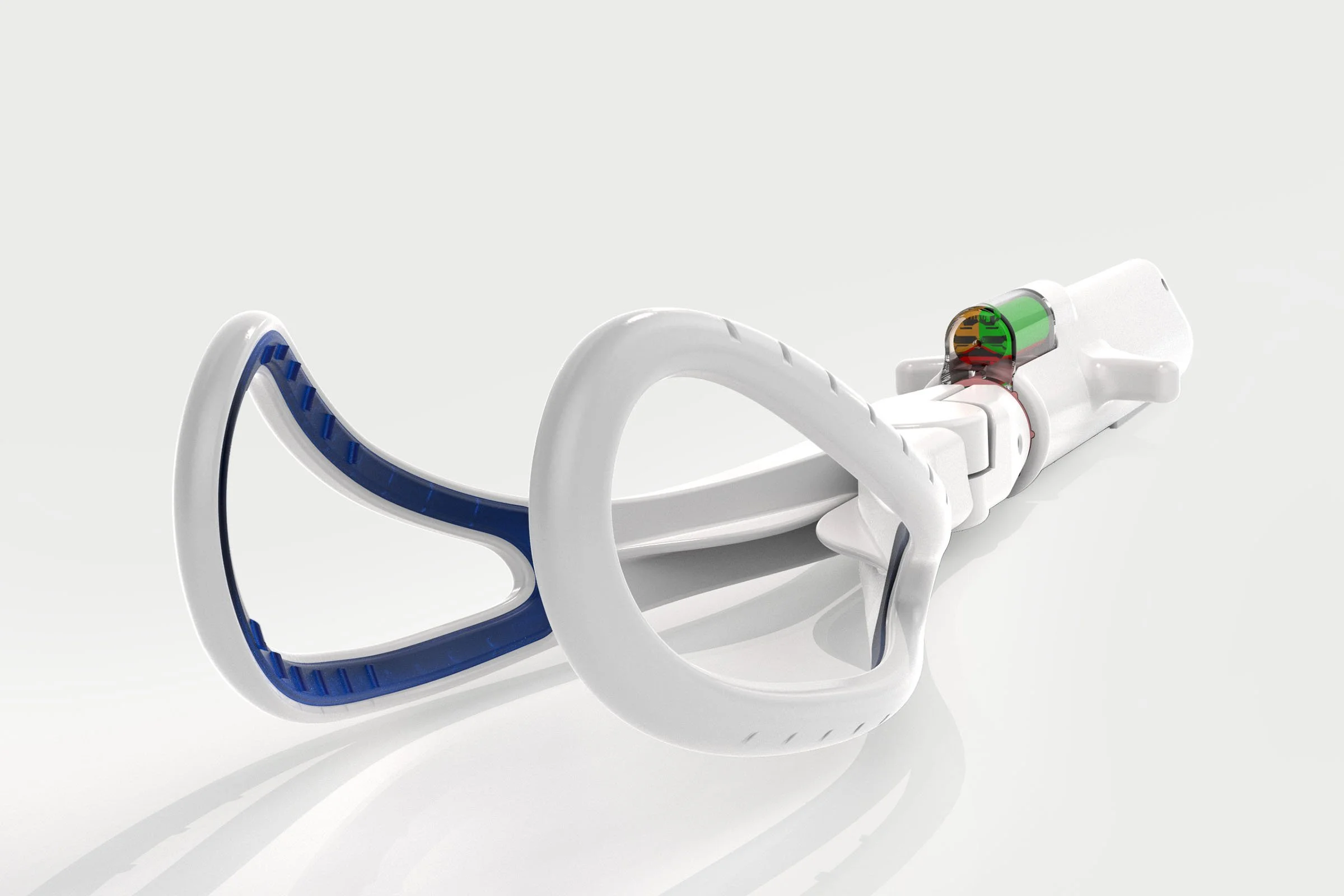
Approach: Working closely with the clinical team we supported anatomical modelling and mannequin-based studies to understand safe traction ranges during assisted delivery. Insights informed development of a redesigned forceps concept incorporating improved grip characteristics and an integrated mechanical load-measurement feature within the handle. We progressed the solution through a full product development cycle, exploring materials, sensing approaches, ergonomics and manufacturability to create a robust, clinically relevant instrument suitable for further commercialisation.
Challenges: A key challenge was addressing a critical safety issue where little published guidance existed on safe traction force ranges. The device also needed to integrate a reliable measurement mechanism without compromising familiarity, balance or usability. Achieving meaningful feedback within a traditionally simple surgical instrument required careful engineering and iterative clinical evaluation.
Deliverables: We delivered concept development, ergonomic refinement and engineering design for a next-generation obstetric forceps incorporating a mechanical traction-force indicator. We supported anatomical testing studies, materials selection and technology evaluation, enabling protection through design right and patent application. The resulting design formed the basis of a licensable product ready for transfer to a specialist medical device manufacturer.
Impact: The project introduced measurable traction guidance to a centuries-old instrument, supporting safer assisted delivery and improved clinical confidence. Our development work enabled IP protection and helped accelerate transfer to a commercial manufacturer, shortening downstream development timelines and supporting translation toward real-world clinical use.



“The Pd-m team worked really closely with us and were an integral part of the project team. They really understood the brief and what the product needed to do, producing a design that allowed us to file both design right and patent applications. We have subsequently licensed the IP to a medical device company for manufacture and the work that Pd-m did has certainly shortened their development timescales.”
